R. Gao, M. Beladi-Mousavi, G. Salinas, L. Zhang, A. Kuhn
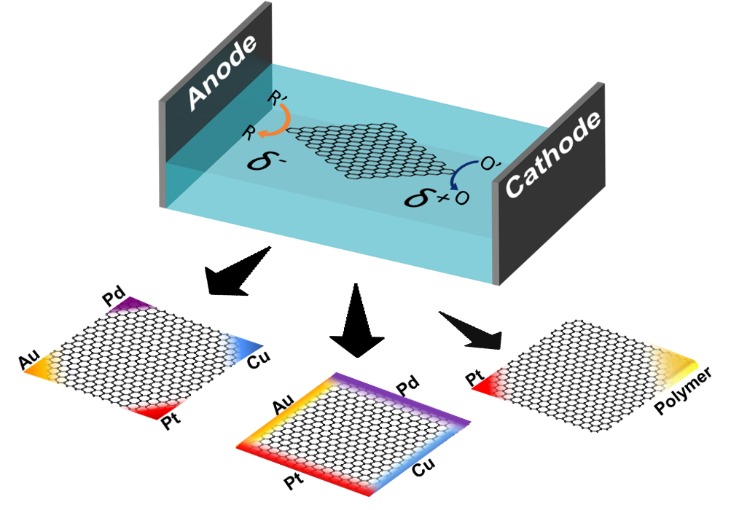
Graphene has gained substantial research interest in many fields due to its remarkable properties among many other two-dimensional materials. In this study, we propose a wireless electrochemical approach, bipolar electrochemistry, for the precise modification of single layers of graphene at predefined locations, such as distinct edges or corners, with a variety of metals or polymers, thus enabling the elaboration of multi-functional monolayer graphene sheets. We illustrate the concept e.g. by depositing multiple metals, or platinum and a catalyst-containing porous polymer on the same graphene sheet, but at separate corners. This configuration allows activating chemiluminescence on the polymer spot, and simultaneously generates the driving force for autonomous motion on the Pt side through the catalytic decomposition of hydrogen peroxide into oxygen bubbles. This integration of different chemical features on the same object, exemplified by these proof-of-principle experiments, enhances the functionality of two-dimensional materials, paving the way for the use of these hybrid materials for a variety of applications, ranging from sensing and catalysis to targeted delivery.
Yiran Zhao, Borja Sépulveda, Julie Descamps, Fatoumata Faye, Marcos Duque, Jaume Esteve, Lionel Santinacci, Neso Sojic, Gabriel Loget, Yoan Léger
ACS Appl. Mater. Interfaces 2024, 16, 9, 11722–11729
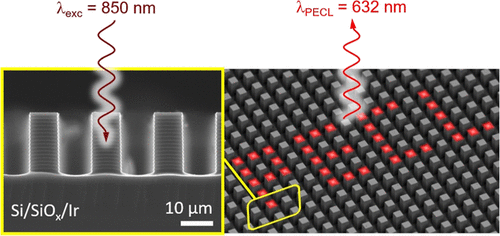
Infrared (IR) imaging devices that convert IR irradiation (invisible to the human eye) to a visible signal are based on solid-state components. Here, we introduce an alternative concept based on light-addressable electrochemistry (i.e., electrochemistry spatially confined under the action of a light stimulus) that involves the use of a liquid electrolyte. In this method, the projection of a near-IR image (λexc = 850 or 840 nm) onto a photoactive Si-based photoanode, immersed into a liquid phase, triggers locally the photoinduced electrochemiluminescence (PECL) of the efficient [Ru(bpy)3]2+-TPrA system. This leads to the local conversion of near-IR light to visible (λPECL = 632 nm) light. We demonstrate that compared to planar Si photoanodes, the use of a micropillar Si array leads to a large enhancement of local light generation and considerably improves the resolution of the PECL image by preventing photogenerated minority carriers from diffusing laterally. These results are important for the design of original light conversion devices and can lead to important applications in photothermal imaging and analytical chemistry.
J. Dabboussi, R.-A. Eichel, H. Kungl, R. Abdallah, G. Loget*
Curr. Opin. Electrochem., 2024, 45, 101468.
Despite the longstanding interest in urea oxidation reaction (UOR), the identification of reaction products under conventional conditions was only reported recently. It turns out that the initially thought “sustainable pathway”, leading to harmless products, represents just a small fraction of the overall reaction mechanism. This is detrimental as the use of urea-rich aqueous feeds for H2 production, along with their remediation through UOR, constitutes perhaps the most important added value of this process for power-to-X and clinical applications. Nonetheless, promising strategies favoring the formation of environmentally friendly products over harmful overoxidized ones already exist. This is expected to lead to a “rebirth” of this research field and open the quest for ultimate selectivity to ensure the complete sustainability of UOR. Therefore, the systematic analysis of reaction products, the elucidation of mechanisms for improving N2 faradaic efficiency, and the design of selective catalysts should be the next focus of research in the field of UOR.

Y. Zhao, Y. Léger, J. Descamps, N. Sojic, G. Loget
Electrochemiluminescence (ECL) is the generation of light induced by an electrochemical reaction, driven by electricity. Here, an all-optical ECL (AO–ECL) system is developped, which triggers ECL by the illumination of electrically autonomous “integrated” photoelectrochemical devices immersed in the electrolyte. Because these systems are made using small and cheap devices, they can be easily prepared and readily used by any laboratories. They are based on commercially available p-i-n Si photodiodes (≈1 € unit−1), coupled with well-established ECL-active and catalytic materials, directly coated onto the component leads by simple and fast wet processes. Here, a Pt coating (known for its high activity for reduction reactions) and carbon paint (known for its optimal ECL emission properties) are deposited at cathode and anode leads, respectively. In addition to its optimized light absorption properties, using the commercial p-i-n Si photodiode eliminates the need for a complicated manufacturing process. It is shown that the device can emit AO–ECL by illumination with polychromatic (simulated sunlight) or monochromatic (near IR) light sources to produce visible photons (425 nm) that can be easily observed by the naked eye or recorded with a smartphone camera. These low-cost off-grid AO-ECL devices open broad opportunities for remote photodetection and portable bioanalytical tools.

Y. Boukarkour, S. Reculusa, N. Sojic, A. Kuhn, G. Salinas
Chem.-Eur. J. (2024) e202400078
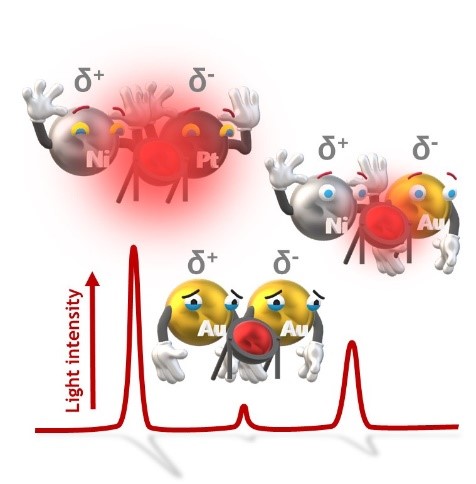
Water splitting has become a sustainable and clean alternative for hydrogen production. Commonly, the efficiency of such reactions is intimately related to the physico-chemical properties of the catalysts that constitute the electrolyzer. Thus, the development of simple and fast methods to evaluate the electrocatalytic efficiency of an electrolyzer is highly required. In this work, we present an unconventional method based on the combination of bipolar electrochemistry and light-emitting diodes, which allows the evaluation of the electrocatalytic performance of the two types of catalysts, composing an electrolyzer, namely for oxygen and hydrogen evolution reactions. The integrated light emission of the diode acts as an optical readout of the electrocatalytic information, which simultaneously depends on the composition of the anode and the cathode. The electrocatalytic activity of Au, Pt and Ni electrodes, connected to the LED in multiple anode/cathode configurations, towards the water splitting reactions has been evaluated. The efficiency of the electrolyser can be represented in terms of the onset electric field (εonset) for light emission, obtaining variations that are in agreement with data reported with conventional electrochemistry. This work introduces a straightforward method for evaluating electrocatalysts and underscores the importance of material characterization in developing efficient electrolyzers for hydrogen production.
- Fine-Tuning the optoelectronic and redox properties of an electropolymerized thiophene derivative for highly selective OECT-based zinc detection
- Infrared photoinduced electrochemiluminescence microscopy of single cells
- All-Optical Electrochemiluminescence at Metal-Insulator-Semiconductor Diodes
- Photoelectrode/electrolyte interfacial band lineup engineering with alloyed III–V thin films grown on Si substrates






