L. Digel, M. Mierzwa, R. Bonné, S. E. Zieger, I.-A. Pavel, E. Ferapontova, K. Koren, T. Boesen, F. Harnisch, I. P. G. Marshall, L. P. Nielsen, A. Kuhn
Angew. Chem. Int. Ed. (2023) e202312647
see also Highlight in Nature Reviews Chemistry
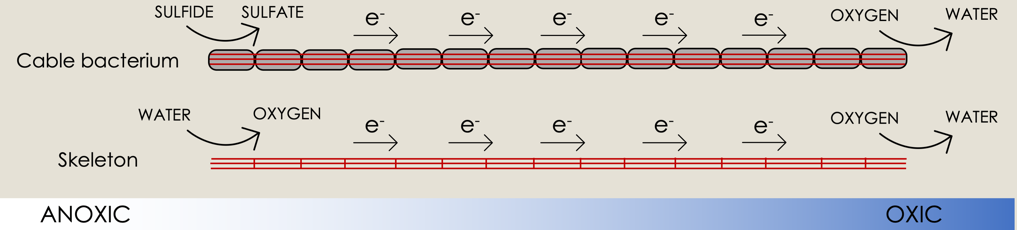
Cable bacteria are multicellular, filamentous bacteria that use internal conductive fibers to transport electrons over centimeter distances from donors within anoxic sediment layers to oxygen at the surface. We extracted the fibers and used them as free-standing bio-based electrodes to investigate their electrocatalytic behavior. The fibers catalyzed the reversible interconversion of oxygen and water, and an electric current was running through the fibers even when the potential difference was generated solely by a gradient of oxygen concentration. Oxygen reduction as well as oxygen evolution were confirmed by optical measurements. Within living cable bacteria, oxygen reduction by direct electrocatalysis on the fibers and not by membrane-bound proteins readily explains exceptionally high cell-specific oxygen consumption rates observed in the oxic zone, while electrocatalytic water oxidation may provide oxygen to cells in the anoxic zone.
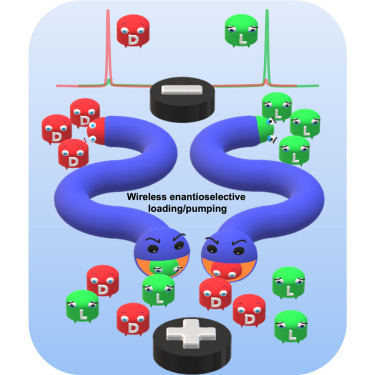
S. Grecchi, G. Salinas, R. Cirilli, T. Benincori, S. Ghirardi, A. Kuhn, S. Arnaboldi
Chirality plays a crucial role in different research fields, ranging from fundamental physico-chemistry to applied aspects in materials science and medicine. In this context, enantioselective loading and pumping of chiral analytes for analysis, separation and cargo delivery applications is an interesting scientific challenge. Herein we introduce artificial chiral soft electromechanical pumps based on a bi-layer film, built up by electrodepositing polypyrrole and an inherently chiral conducting oligomer at its internal surface. The enantioselective device can be driven by bipolar electrochemistry to act as a pump, allowing the selective loading and separation of different chiral analytes injected as pure enantiomers and in their racemic form (i.e., doxorubicin, a chemotherapy drug, limonene, carvone and a chiral ferrocene). The synergy between the wireless electromechanical actuation and the inherent enantiodiscrimination features makes these soft actuators excellent candidates for the controlled handling of chiral molecules in the frame of potential applications ranging from analysis to drug delivery.
K. Tieriekhov, N. Sojic, L. Bouffier, G. Salinas, A. Kuhn
Adv. Science 11 (2024) 2306635
Electromagnetically induced rotation is a key process of many technological systems that are used in daily life, especially for energy conversion. In this context, the Lorentz force-induced deviation of charges is a crucial physical phenomenon to generate rotation. Herein, we combine the latter with the concept of bipolar electrochemistry to design a wireless magnetoelectrochemical rotor. Such a device can be considered as a wet analogue of a conventional electric motor. The main driving force that propels this actuator is the result of the synergy between the charge-compensating ion flux along a bipolar electrode and an external magnetic field applied orthogonally to the surface of the object. The trajectory of the wirelessly polarized rotor can be controlled by the orientation of the magnetic field relative to the direction of the global electric field, producing a predictable clockwise or anticlockwise motion. Fine-tuning of the applied electric field allows addressing conducting objects having variable characteristic lengths.

M. Sentic, I. Trajkovic, D. Manojlovic, D. Stankovic, M. Vesna Nikolic, N. Sojic and J. Vidic
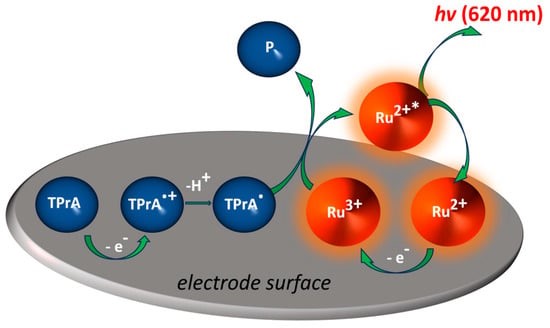
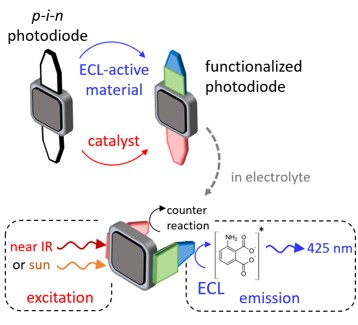
Y. Zhao, Y. Léger, J. Descamps, N. Sojic, G. Loget
Electrochemiluminescence (ECL) is the generation of light induced by an electrochemical reaction, driven by electricity with a power supply. It is a powerful analytical technique that is ubiquitous in immunoassays and clinical diagnosis. Here, we develop an all-optical ECL (AO-ECL) system, which triggers ECL by the illumination of electrically-autonomous “integrated” photoelectrochemical devices immersed in the electrolyte. Because these systems are made using small and cheap devices, they can be easily prepared and readily used by any laboratories. They are based on commercially available p-i-n Si photodiodes (~1 €/unit), coupled with well-established ECL-active and catalytic materials, directly coated onto the component leads by simple and fast wet processes. Here, a Pt coating (known for its high activity for reduction reactions) is deposited at cathode lead, and, carbon paint (known for its optimal ECL emission properties) is deposited at the anode lead. In addition to its optimized light absorption properties, using the commercial p-i-n Si photodiode eliminates the need for a complicated manufacturing process. We show that the device can emit AO-ECL by illumination with polychromatic (simulated sunlight) or monochromatic (near IR) light sources to produce visible photons (425 nm) that can be easily observed by the naked eye or recorded with a smartphone camera. These low-cost off-grid AO-ECL devices open broad opportunities for remote photodetection and portable bioanalytical tools.
- Optics determines the electrochemiluminescence signal of bead-based immunoassays
- Endogenous and exogenous wireless multimodal light-emitting chemical devices
- Local Reactivity of Metal-Insulator-Semiconductor Photoanodes Imaged by Photoinduced Electrochemiluminescence Microscopy
- Dynamic maping of electrochemiluminescence reactivity in space: application to bead-based assays






