Peyrard L., Dumartin M.-L., Chierici S., Pinet S., Jonusauskas G., Meyrand P., Gosse I.,
J. Org. Chem. 2012, 77, 7023-7027.
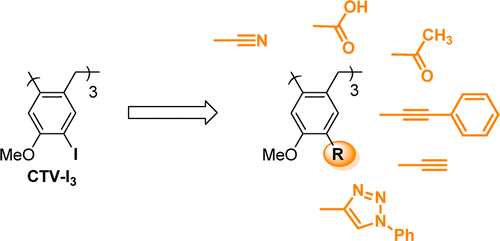
Cyclotriveratrylene analogues (CTVs) are supramolecular bowl-shaped molecules known for their ability to complex organic and organometallic guests, to form liquid crystals, polymers, or nanostructures. In this Article, we report the synthesis of new cyclotriveratrylene analogues with fluorescence properties in which various electron-withdrawing or π-extended conjugated groups are appended to the wide rim ortho to the methoxy-donating groups. Synthetically, these functionalized CTVs cannot be obtained as CTVs with electron-rich functions by the typical method (i.e., the trimerization of the corresponding benzyl alcohol) but are prepared from a common key intermediate, the C3-triiodocyclotriveratrylene (CTV-I3), in good yields. Despite the synthetic difficulties encountered due to the presence of three reactive centers, we have demonstrated the possibility of performing Sonogashira coupling and Huisgen cycloaddition reactions directly to the CTV core for the first time. CTVs with π-extended conjugated groups reveal interesting fluorescence profiles. More broadly, this study utilizes CTV-I3 to introduce novel functionalities into CTVs to keep exploring their potential applications.
Bouffier L., Gosse I., Demeunynck M., Mailley P.,
Bioelectrochemistry 2012, 88, 103-109.
We report here the electrochemical characterization of eight synthetic DNA intercalators based on the 4H-pyrido[4,3,2-kl]acridin-4-one structure. We found that the electrochemical behavior of these redox active drugs is strongly influenced by the nature of the solvent. A single two-electron reduction is observed in an aqueous phosphate buffer (PB) whereas two successive one-electron reductions are observed in aprotic solution (acetonitrile). The influence of the molecular structure on the potential values is addressed along with a comparison between the DNA binding constant (KDNA) and the cytotoxic activity against HT29 cells (IC50). For typical DNA intercalators, one could expect that toxicity will be roughly proportional to the DNA binding constant. Yet, a structure/activity comparison solely based on the DNA affinity was not conclusive. In contrast, a direct relationship was evidenced for the first time between the decimal logarithm of the in vitro bioactivity and the reduction potential of pyridoacridones recorded in PB at pH 7.0. Moreover, most of the bio/electrochemical relationships previously described for quinone-based drugs were reported with electrochemical characterization in aprotic solvents (typically acetonitrile, dimethylformamide or dimethylsulfoxide). But aqueous solution electrochemistry is definitely the most bio-relevant because the redox mechanism of quinone or iminoquinone reduction directly depends on the protic nature of the solvent.
Loget G., Roche J., Kuhn A.,
Adv. Mater. 2012, 24, 5111-5116. Cover article
Janus particles have different features/chemistry on two opposite sides (see figure). So far, they have been obtained mainly by two-dimensional synthetic methods, which are able to break the symmetry but limit the amount of produced particles. A true bulk approach, based on bipolar electrochemistry, is presented that allows the straightforward synthesis of such asymmetric micro- and nano-objects.
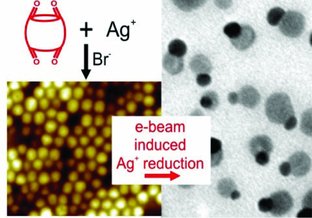
Loget G., Lee T.-C., Taylor R. W., Mahajan S., Nicoletti O., Jones S. T., Coulston R. J., Lapeyre V., Garrigue P., Midgley P. A., Scherman O. A., Baumberg J. J., Kuhn A.,
Small 2012, 8, 2698-2703.
The straight-forward synthesis of Janus nanoparticles composed of Ag and AgBr is reported. For their formation, cucurbit[n]uril (CB)-stabilized AgBr nanoparticles are first generated in water by precipitation. Subsequent irradiation with an electron beam transforms a fraction of each AgBr nanoparticle into Ag0, leading to well-defined Janus particles, stabilized by the binding of CB to the surface of both AgBr and Ag0. With the silver ion reduction being triggered by the electron beam, the progress of the transformation can be directly monitored with a transmission electron microscope.
Heim M., Rousseau L., Reculusa S., Urbanova V., Mazzocco C., Joucla S., Bouffier L., Vytras K., Bartlett P., Kuhn A., Yvert B.
Journal of Neurophysiology 2012, 15, 1793-1803.
Microelectrode arrays (MEAs) are appealing tools to probe large neural ensembles and build neural prostheses. Microelectronics microfabrication technologies now allow building high-density MEAs containing several hundreds of microelectrodes. However, several major problems become limiting factors when the size of the microelectrodes decreases. In particular, regarding recording of neural activity, the intrinsic noise level of a microelectrode dramatically increases when the size becomes small (typically below 20-μm diameter). Here, we propose to overcome this limitation using a template-based, single-scale meso- or two-scale macro-/mesoporous modification of the microelectrodes, combining the advantages of an overall small geometric surface and an active surface increased by several orders of magnitude. For this purpose, standard platinum MEAs were covered with a highly porous platinum overlayer obtained by lyotropic liquid crystal templating possibly in combination with a microsphere templating approach. These porous coatings were mechanically more robust than Pt-black coating and avoid potential toxicity issues. They had a highly increased active surface, resulting in a noise level ∼3 times smaller than that of conventional flat electrodes. This approach can thus be used to build highly dense arrays of small-size microelectrodes for sensitive neural signal detection.
- Bulk Synthesis of Janus Objects and Asymmetric Patchy Particles
- Bipolar Electrochemistry for Cargo-lifting in Fluid Channels
- Origin and Control of Adhesion between Emulsion Drops Stabilised by Thermally-sensitive Soft Colloidal Particles
- Site Selective Generation of Sol-gel Deposits in Layered Bi-metallic Macroporous Electrode Architectures