C. Adam, A. Wallabregue, H. Li, J. Gouin, R. Vanel, S. Grass, J. Bosson, L. Bouffier, J. Lacour and N. Sojic.
Chem. Eur. J., 2015, 21, 19243-19249.
Trianguleniums are fascinating conjugated hexacyclic cations that exhibit interesting electronic and optical properties. Herein, the electrogenerated chemiluminescence (ECL) emission of this family of fluorescent dyes is reported for the first time. Redox behavior and fluorescence properties of eight cationic triangulene luminophores with different heteroatom patterns in the core structure and various pending substituents were examined to rationalize the ECL. Clearly, the more electron-rich the carbocation, the more efficient the corresponding ECL; two very distinct classes of triangulenes can be drawn from these studies by using an ECL wall sufficiency formalism.

S. Chen, G. Wantz, L. Bouffier and J. Gao.
ChemElectroChem, 2016, 3, 392-398.
We present solid-state, polymer-based, light-emitting electrochemical cells incorporating bipolar electrodes (BPEs) positioned between the driving electrodes. The dominant processes in these planar electrochemical cells are in situ electrochemical p and n doping of the luminescent conjugated polymer when a sufficient voltage bias is applied between the feeder anode and cathode. The inclusion of floating BPEs causes additional doping at the extremities of the BPE, which has been visualized through in situ fluorescence imaging. We investigate how the applied voltage bias and the BPE size affect electrochemical doping from the BPE. We observe an interesting delay in the appearance of doping from the BPE if the applied voltage bias was initially insufficient. Doping eventually occurs when the potential difference across the BPE/polymer interface becomes sufficient, as doping from the driving electrodes propagates. The inclusion of BPEs can lead to more efficient polymer light-emitting and photovoltaic cells when a large number of active junctions are formed.

E. Pedraza, A. Karajić, M. Raoux, R. Perrier, A. Pirog, F. Lebreton, S. Arbault, J. Gaitan, S. Renaud, A. Kuhn, J. Lang
We are developing a cell-based bioelectronic glucose sensor that exploits the multi-parametric sensing
ability of pancreatic islet cells for the treatment of diabetes. These cells sense changes in the concentration
of glucose and physiological hormones and immediately react by generating electrical signals. In our sensor,
signals from multiple cells are recorded as field potentials by a micro-electrode array (MEA). Thus, cell
response to various factors can be assessed rapidly and with high throughput. However, signal quality and
consequently overall sensor performance rely critically on close cell–electrode proximity. Therefore, we
present here a non-invasive method of further exploiting the electrical properties of these cells to guide
them towards multiple micro-electrodes via electrophoresis. Parameters were optimized by measuring the
cell's zeta potential and modeling the electric field distribution. Clonal and primary mouse or human
β-cells migrated directly to target electrodes during the application of a 1 V potential between MEA
electrodes for 3 minutes. The morphology, insulin secretion, and electrophysiological characteristics were
not altered compared to controls. Thus, cell manipulation on standard MEAs was achieved without introducing
any external components and while maintaining the performance of the biosensor. Since the analysis
of the cells' electrical activity was performed in real time via on-chip recording and processing, this work
demonstrates that our biosensor is operational from the first step of electrically guiding cells to the final
step of automatic recognition. Our favorable results with pancreatic islets, which are highly sensitive and
fragile cells, are encouraging for the extension of this technique to other cell types and microarray devices.

M. Valetaud, G. Loget, J. Roche, N. Hüsken, Z. Fattah, V. Badets, O. Fontaine, D. Zigah.
J. Chem. Educ., 2015, 92 (10), pp 1700–1704.
The Electrochemical Pen (EChemPen) was developed as an attractive tool for learning electrochemistry. The fabrication, principle, and operation of the EChemPen are simple and can be easily performed by students in practical classes. It is based on a regular fountain pen principle, where the electrolytic solution is dispensed at a tip to locally modify a conductive surface by triggering a localized electrochemical reaction. Three simple model reactions were chosen to demonstrate the versatility of the EChemPen for teaching various electrochemical processes. We describe first the reversible writing/erasing of metal letters, then the electrodeposition of a black conducting polymer “ink”, and finally the colorful writings that can be generated by titanium anodization and that can be controlled by the applied potential. These entertaining and didactic experiments are adapted for teaching undergraduate students that start to study electrochemistry by means of surface modification reactions.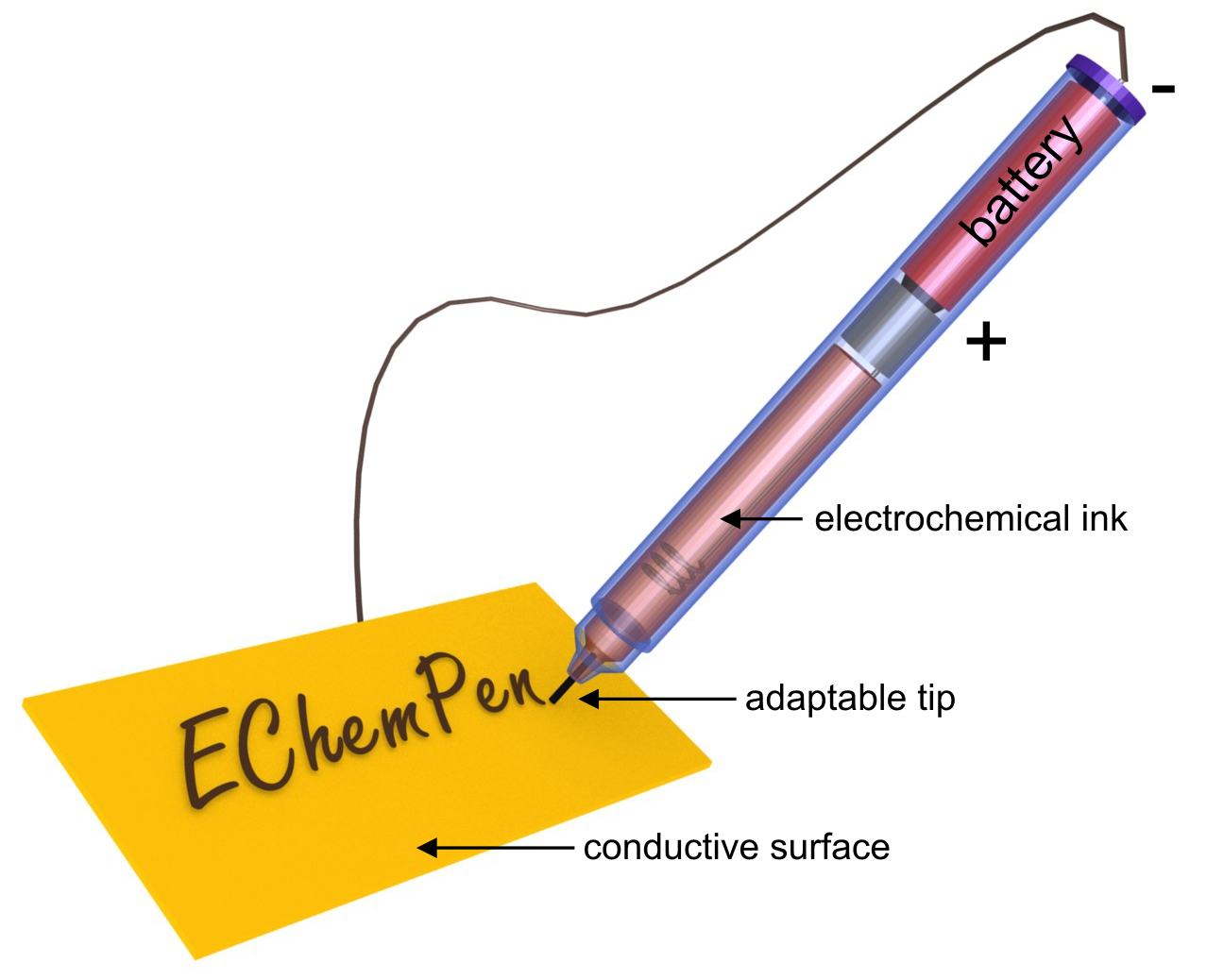
C. Kumsapaya, J. Limtrakul, A. Kuhn, D. Zigah, C. Warakulwit.
ChemElectroChem, 2016, 3, 410-414.
Vertically aligned carbon nanotubes were grown over the inner wall of the pores of an anodic aluminum oxide template via chemical vapor deposition. The versatile approach of bipolar electrochemistry was generalized to modify the tubes selectively on one end with an organic layer of 4-aminobenzene moieties. The presence of the template during the bipolar experiments intrinsically protects the tubes' outer surface from the grafting of the organic layer. The layer is thus not only selectively grafted at the cathodic side of the tubes, but also exclusively onto the tubes' inner surface. For longer deposition times, the end of the tubes was completely capped with a dense organic layer. Thus bipolar electrochemistry can not only generate CNTs bearing organic functionalities on the tube surface for bio-sensing or bio-electrocatalytic applications, but can also create a dense organic layer as a cap at one end of the tubes, opening up applications in the fields of drug storage and delivery.
- Single-Step Screening of the Potential Dependence of Metal Layer Morphologies Along Bipolar Electrodes
- Enantioselective Recognition of DOPA by mesoporous Platinum imprinted with Mandelic acid
- Bottom-up Generation of Miniaturized Coaxial Double Electrodes with tunable Porosity
- 3D electrogenerated chemiluminescence: from surface-confined reactions to bulk emission






